IV Fluids 101: Types, Uses, and What They Actually Do
IV Fluids 101: Types, Uses, and What They Actually Do
Joseph Lopez • June 2026 Medically reviewed by Micaela Strevay, FNP-C, PMHNP-BC.
This guide is for general health information. It is not a substitute for medical advice. If you have chest pain, confusion, trouble breathing, or signs of severe dehydration, call 911 or visit urgent care.
You're sitting in a hospital bed after a rough bout of food poisoning. A nurse hooks a clear bag to a pole and runs a thin tube to your arm. "This will help," she says. But what is in that bag? Why that one? And why can't you just drink water?
This guide answers all of it.
Quick answer: IV fluids are sterile liquid solutions delivered directly into your vein to restore fluid, electrolytes, or both. There are several types, each designed for a different job. The right one depends on what your body is missing, how fast you need it corrected, and what condition you have. A licensed nurse or doctor selects the fluid that fits your situation.

What Are IV Fluids?
Your body is about 60% water ( Cleveland Clinic ). That water carries electrolytes like sodium, potassium, and chloride through every cell and organ. When you get sick, lose blood, or stop eating and drinking, those levels drop.
IV fluids (short for intravenous fluids) go straight into a vein, bypassing your stomach and intestines entirely. The fluid is in your bloodstream within seconds of entering the IV line. About 25 million people receive IV therapy every year, making it the most common invasive medical procedure in the world ( PMC ).
Clinicians use IV fluids for three main purposes ( StatPearls: Fluid Management, NIH ):
- Maintenance: meeting your daily fluid needs when you can't eat or drink.
- Replacement: correcting a specific deficit (lost blood, lost electrolytes, dehydration).
- Resuscitation: quickly restoring blood volume in emergencies like trauma or sepsis.
IV Fluids vs. Drinking Water: Why the Drip Works Faster
This is one of the most-searched questions about IV therapy, and none of the major medical sites actually answer it. Here is the plain version.
How water absorption works
When you drink water, it goes into your stomach. Then it moves into your small intestine, where most absorption happens. From there, it crosses into your bloodstream. The whole process takes 45 minutes to several hours, depending on how much is in your stomach, how well your gut is working, and what else you've eaten ( Destination Hydration IV ).
If you're vomiting or have diarrhea, that process is even slower. Sometimes it doesn't work at all. You throw up the water before it can absorb.
How IV bypasses all of that
An IV line puts fluid directly into your vein. There is no digestion step. Bioavailability is 100%. Your cells start getting what they need within minutes. Most people feel the effects (reduced headache, less dizziness, more energy) before the bag is even halfway done.
A standard 1-liter bag typically runs over 30 to 60 minutes at a normal infusion rate ( NCBI Nursing Skills ). Drink a liter of water in that same 30 minutes and your body won't fully absorb it for another hour or two.
The short answer: IV wins on speed, every time. If your gut is working fine and you have time, drinking water is cheaper and carries zero procedural risk. If you're in bad shape, IV is faster and more reliable.
The Two Main Categories: Crystalloids and Colloids
Before getting into specific fluid types, you need to know the two big buckets.
Crystalloids
Crystalloids are water-based solutions with dissolved salts, sugars, or both. They flow freely across cell membranes. They're inexpensive, widely available, and make up the vast majority of IV fluids. Normal saline, lactated Ringer's, Plasmalyte, D5W, and half normal saline are all crystalloids.
Colloids
Colloids contain larger molecules (proteins, starches, or synthetic compounds) that stay in the bloodstream longer because they can't cross vessel walls as easily. Examples are albumin and dextran.
A Cochrane Review of 21,000+ patients found colloids make "little or no difference" in mortality compared to crystalloids, but cost significantly more and carry a higher allergy risk. Crystalloids are the standard of care for most situations.
Tonicity Explained Simply
Every IV fluid has a tonicity: hypotonic, isotonic, or hypertonic. This describes how the fluid affects your cells.
Think of your cells as tiny water balloons. The fluid inside them has a certain concentration of dissolved particles. The IV fluid has its own concentration. The relationship between the two determines what happens next.
Osmolarity is the measure of that concentration, expressed as mOsm/L. Normal plasma sits between 275 and 295 mOsm/L ( StatPearls: Crystalloid Solutions, NIH ).
Isotonic: the balanced option
An isotonic fluid has roughly the same osmolarity as your blood plasma. Your cells stay the same size. Fluid moves between compartments without pushing water in or out of cells. Normal saline (~308 mOsm/L), lactated Ringer's (~273 mOsm/L), and Plasmalyte (~294 mOsm/L) are all isotonic.
Hypotonic: pushes water into cells
A hypotonic fluid has fewer dissolved particles than your plasma. Water moves from the fluid into your cells. Cells swell. This is useful for cellular dehydration or high sodium, but can cause dangerous brain swelling if used incorrectly. Half normal saline (~154 mOsm/L) is the most common hypotonic fluid.
Hypertonic: pulls water out of cells
A hypertonic fluid has more dissolved particles than your plasma. Water leaves your cells and moves into the fluid. Cells shrink. Blood volume increases. Hypertonic fluids like D5½NS (~406 mOsm/L) are reserved for specific emergencies: cerebral edema, severe hyponatremia, or very low blood pressure. Always given in monitored settings.
The Most Common Types of IV Fluids
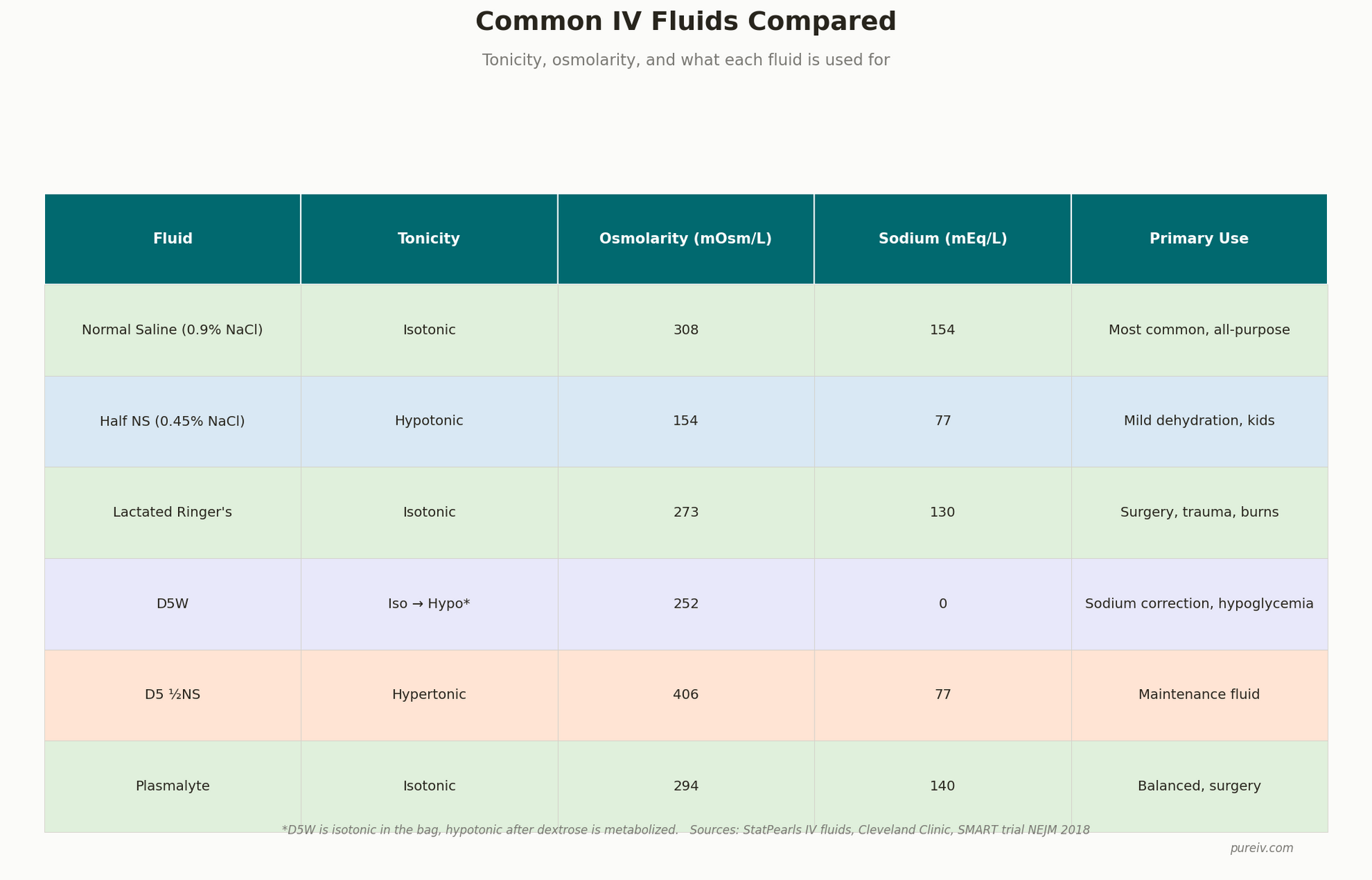
| Fluid | Tonicity | Osmolarity | Primary Use |
|---|---|---|---|
| Normal Saline (0.9% NaCl) | Isotonic | ~308 mOsm/L | Hydration, blood transfusions, vomiting/diarrhea |
| Half Normal Saline (0.45% NaCl) | Hypotonic | ~154 mOsm/L | Cellular dehydration, hypernatremia |
| Lactated Ringer's | Isotonic | ~273 mOsm/L | Trauma, burns, surgery, metabolic acidosis |
| D5W (5% Dextrose in Water) | Isotonic → Hypotonic* | ~252 mOsm/L | Hypernatremia, electrolyte dilution, dextrose delivery |
| Plasmalyte | Isotonic | ~294 mOsm/L | Balanced resuscitation, closest to plasma |
| D5½NS | Hypertonic | ~406 mOsm/L | Severe hyponatremia, cerebral edema |
D5W becomes hypotonic once your body metabolizes the dextrose, leaving behind only free water.
Normal Saline (0.9% NaCl): the universal default
Normal saline is the most used IV fluid in the world. It's a saline drip: just water and salt. Specifically, it's 0.9% sodium chloride dissolved in sterile water.
Its osmolarity is about 308 mOsm/L, which places it just above normal plasma range ( StatPearls: Crystalloid Solutions, NIH ). Technically that makes it slightly hypertonic, but it's classified and used as isotonic for most clinical purposes.
Normal saline works for almost everything: dehydration from vomiting or diarrhea, low blood volume, blood transfusions, and as a base for IV medications.
The downside: normal saline has much more chloride than your blood does. It contains 154 mmol/L vs. your plasma's ~100 mmol/L. Giving large amounts can cause a condition called hyperchloremic metabolic acidosis, where your blood becomes more acidic than it should be. This matters more in hospital settings where large volumes are used. For a single wellness infusion, the risk is very low.

Lactated Ringer's: the balanced choice
Lactated Ringer's (LR) is considered more balanced than normal saline. Its composition per liter: sodium 130 mmol/L, chloride 109 mmol/L, potassium 4 mmol/L, calcium 1.5 mmol/L, and lactate 28 mmol/L ( StatPearls: Crystalloid Solutions, NIH ).
Those numbers are much closer to what's actually in your blood plasma. The lactate is converted to bicarbonate by your liver, which actually helps correct mild acidosis rather than worsen it ( Healthline ).
LR is commonly used in surgery, trauma, burn treatment, and whenever large volumes need to be given. It's also the base fluid in many wellness IV drips, including Myers' Cocktail.
There are situations where you wouldn't use LR. If you have liver disease, your liver can't process the lactate. If you have lactic acidosis (already too much lactate in your blood), adding more is a problem. LR also has calcium, which means it doesn't mix well with certain medications, including medications given via blood transfusion ( WebMD ).
Your nurse will ask about your medical history before choosing LR. That question isn't small talk.
Plasmalyte: the most plasma-like option
Most people outside of hospitals have never heard of Plasmalyte, but it deserves its own section.
Plasmalyte 148 is a balanced crystalloid with a pH of about 7.4, the same as healthy human blood. Its osmolarity is approximately 294 mOsm/L. It contains sodium, potassium, magnesium, chloride, acetate, and gluconate ( PMC: Plasma-Lyte 148 Clinical Review ).
No other commonly used IV fluid matches human plasma this closely. The absence of lactate (replaced by acetate and gluconate) means it can be used even in patients with liver problems, unlike LR. It doesn't promote the acidosis that large volumes of normal saline can cause.
Plasmalyte is increasingly used in surgery and critical care. It was one of the balanced crystalloids in the landmark SMART trial (more on that below).
D5W (5% Dextrose in Water): the sugar water that isn't what it seems
D5W starts as an isotonic fluid with an osmolarity of about 252 mOsm/L. But once your body metabolizes the dextrose (sugar), what's left is free water. At that point it behaves like a hypotonic fluid.
This matters because free water distributes everywhere, including into brain cells. That's why D5W should never be used for fluid resuscitation or large-volume hydration in most patients. Doing so risks cerebral edema ( NCBI Nursing Skills: Comparison of IV Solutions ).
D5W's appropriate uses are specific: delivering dextrose to a patient with low blood sugar, treating high sodium (hypernatremia) by diluting the sodium concentration, or serving as a vehicle for certain IV medications that need a dextrose solution.
Half Normal Saline (0.45% NaCl): for cellular dehydration
Half normal saline is exactly what it sounds like: normal saline cut in half. Osmolarity is about 154 mOsm/L, which is hypotonic.
Because it has fewer particles than your plasma, water moves from the fluid into your cells. This makes it useful when cells are dehydrated even though blood volume is okay. That situation is called hypernatremia (too much sodium in the blood). Bringing that sodium down requires delivering water into the cells, not just the bloodstream.
Half normal saline is not appropriate for patients with liver disease, head trauma, or burns, where brain swelling is a risk ( NCBI Nursing Skills: Comparison of IV Solutions ).
D5NS and D5½NS: hypertonic combinations
These fluids combine dextrose with normal saline (D5NS) or half normal saline (D5½NS). The addition of dextrose pushes osmolarity above plasma levels. D5½NS sits around 406 mOsm/L, firmly in hypertonic territory.
Hypertonic fluids pull water out of cells and into the bloodstream. They're used in specific medical emergencies: severe low sodium, cerebral edema, or situations where increasing blood osmolarity quickly is the clinical goal. These are always given in a monitored hospital or clinical setting. You would not receive these at a mobile IV clinic.
What the Science Says: The SMART Trial
Here's where it gets interesting for anyone who thought normal saline was always the safest default.
In 2018, the NEJM published the SMART trial: 15,802 critically ill adults at Vanderbilt University Medical Center received either normal saline or a balanced crystalloid (LR or Plasmalyte). Balanced crystalloids reduced major adverse kidney events to 14.3% vs. 15.4% with saline (OR 0.91, P=0.04). In-hospital mortality was 10.3% vs. 11.1% with saline ( SMART Trial, NEJM 2018 ).
For most healthy adults getting a single wellness infusion, the difference between NS and LR is not dramatic. But for a nurse managing large-volume therapy in a sick patient, the balanced option is increasingly the standard of care. More hospitals and mobile IV services are moving toward LR and Plasmalyte over plain normal saline.
Real-World Uses: When Do You Actually Get an IV?
Dehydration from illness
Vomiting, diarrhea, and fever all cause fluid and electrolyte loss quickly. If you can't keep water down, an IV is often the only effective option. Most hospitals and mobile IV services use isotonic crystalloids (normal saline or LR) for this. Pure IV's dehydration IV is designed exactly for this situation.
Hangover recovery
Alcohol is a diuretic. It makes your kidneys flush water, leaving you down a liter or more of fluid plus depleted electrolytes and B vitamins. The classic treatment is a banana bag: normal saline or LR plus thiamine, multivitamins, and folate.
Pure IV's hangover IV follows the same logic. The base fluid restores volume and electrolytes; added nutrients address what alcohol strips away. See the banana bag hydration guide and 2-day hangover guide for more.
Surgery and trauma
Large-volume isotonic crystalloids, most often LR or Plasmalyte, are given before, during, and after surgery to maintain blood volume, replace blood loss, and keep organs perfused. Trauma and burn patients need particularly aggressive fluid resuscitation.
Athletic recovery and electrolyte correction
Hard training depletes electrolytes through sweat. A Myers' Cocktail (LR or NS base plus magnesium, B vitamins, and vitamin C) replaces them faster and more completely than sports drinks. See best IV drip for energy for more.
Immune support
Some IV formulas use normal saline or LR as a base for high-dose vitamin C and zinc. Pure IV's Immune Boost IV is built around this concept.
Medication and nutrition delivery
IV antibiotics, chemotherapy, and total parenteral nutrition (TPN) all use saline or dextrose-based fluids as carriers. Your nurse will ask what medications you're on before starting, because LR's calcium can interact with certain drugs.
Risks of IV Fluids
IV therapy is generally safe when a licensed professional administers and monitors it. That said, the risks are real.
Fluid overload
Too much fluid too fast raises blood pressure, causes swelling in the extremities, and in serious cases leads to respiratory failure. Healthy adults with normal kidneys are much less likely to experience this, but it's one reason a nurse should be making the call on volume and rate ( WebMD ).
Electrolyte shifts
The wrong fluid in the wrong situation can push sodium, potassium, or chloride in the wrong direction. Your nurse asks about your health history before selecting a fluid for exactly this reason.
Infection at the IV site
Any time a needle enters a vein, there is a risk of infection. Proper sterile technique keeps this risk very low. Watch for redness, warmth, or swelling at the site after your infusion.
Other risks
Cleveland Clinic lists additional risks: phlebitis (vein inflammation), hematoma, collapsed vein from repeated access, and, rarely, air embolism. These are uncommon with trained practitioners.
Who Can Administer IV Fluids?
In a hospital, IV fluids are given by registered nurses (RNs), physicians, physician assistants, and nurse practitioners. Paramedics can start IVs in emergencies.
Outside the hospital, the rules vary by state. According to Nextech's analysis of IV therapy laws by state , most states require a licensed RN working under physician oversight. Nevada has a more flexible framework for mobile IV clinics. California is stricter.
The U.S. IV hydration market exceeded $1.1 billion in 2025 and is projected to nearly double by 2033 ( Nextech ). That growth has brought in a lot of new providers, some cutting corners. The question of who is legally qualified to give you an IV matters.
At Pure IV, every infusion is performed by a licensed RN who assesses your symptoms and history before selecting a fluid. If you're in a high-altitude city like Denver, Boise, or Salt Lake City, dehydration can hit harder than expected. See the altitude sickness IV guide for more.
How Pure IV Works
Pure IV nurses come to you: home, hotel room, or office, anywhere in Las Vegas, Scottsdale, Phoenix, Denver, Salt Lake City, Dallas, Austin, Albuquerque, Boise, or Nashville.
It isn't a one-size bag pulled off a shelf. The nurse does a brief intake, asks about your symptoms, health history, medications, and allergies, then selects the right fluid and additives. Most infusions take 30 to 60 minutes. Most clients feel better before the bag runs out.
Browse all available Pure IV treatments online.
Frequently Asked Questions
What are the main types of IV fluids?
Crystalloids and colloids are the two main categories. Crystalloids include normal saline, lactated Ringer's, Plasmalyte, D5W, and half normal saline. Each is classified as isotonic, hypotonic, or hypertonic based on how it affects fluid balance in your cells. For most situations, isotonic crystalloids are used.
What is the difference between lactated Ringer's and normal saline?
Both are isotonic crystalloids. Lactated Ringer's is more balanced: its sodium and chloride levels are closer to blood plasma, which reduces the risk of metabolic acidosis with large volumes. Normal saline is compatible with more medications and blood transfusions. Your nurse chooses based on your symptoms and history.
How fast do IV fluids work?
Most people feel effects within minutes. A standard 1-liter bag runs over 30 to 60 minutes. Drinking water takes 45 minutes to several hours to absorb through digestion. If you're vomiting, oral hydration may not work at all.
What IV fluid is given for a hangover?
Usually normal saline or lactated Ringer's plus anti-nausea medication and B vitamins, a combo called a banana bag. It replaces the fluid and electrolytes alcohol's diuretic effect forces out.
Can you get IV fluids at home?
Yes, in many states. Licensed mobile IV services staffed by registered nurses can administer IV therapy at home, in a hotel, or at an office, under physician oversight. Pure IV operates in Las Vegas, Scottsdale, Phoenix, Denver, Salt Lake City, Dallas, Austin, Albuquerque, Boise, and Nashville.
What are the risks of IV fluids?
The main risks are fluid overload (headache, high blood pressure, difficulty breathing), electrolyte imbalances, and infection at the IV site. Less common: phlebitis, hematoma, and, very rarely, air embolism. A licensed nurse monitors for all of these during your infusion.
Book a Pure IV Drip
If you're dehydrated, hungover, run-down, or just want faster recovery, Pure IV can send a licensed nurse to you today. Browse the Pure IV treatments page to see what's available in your city, or book directly online. Most appointments are available same-day.
Sources
- StatPearls: Crystalloid Solutions in Intravenous Fluid Therapy (NIH/NCBI)
- StatPearls: Fluid Management (NIH/NCBI)
- NCBI Nursing Skills: IV Therapy Management, Chapter 23
- NCBI Nursing Skills: Table 15.3 — Comparison of IV Solutions
- SMART Trial: Balanced Crystalloids vs. Saline in Critically Ill Adults — NEJM 2018
- Cochrane Review: Colloids vs. Crystalloids for Fluid Resuscitation
- Plasma-Lyte 148: A Clinical Review (PMC)
- Cleveland Clinic: IV Fluids (Intravenous Fluids): Types & Uses
- WebMD: Lactated Ringer's vs. Normal Saline as IV Fluids
- Healthline: Lactated Ringer's Solution
- Nextech: IV Therapy Laws by State
- Destination Hydration IV: IV Hydration vs. Drinking Water — Which Works Faster?